Contraindication – Do not use Advanced Foot Energizer if
- Powered muscle stimulators should not be used on patients with cardiac demand pacemakers. Do not use if you have a cardiac pacemaker, implanted defibrillators or other implanted electronic device because this may cause electric shock, burns, electrical interference, or death.
- Do not use if you are being treated for, or have the symptoms of, an existing Deep Vein Thrombosis (“DVT”). Symptoms of DVT (blood clot) may include swelling and tenderness in one of your legs (usually calf); heavy ache in the affected area; warm skin in the area of the clot; redness of skin (often at the back of leg below the knee). Consult your doctor if you have signs of DVT symptoms.
- Do not use if your are pregnant as the safety of using powered muscle stimulators during pregnancy has not been established.
- The Advanced Foot Energizer is for use by adults only and is not for children.
Warnings
- The long-term effects of chronic electrical stimulation are unknown.
- Stimulation should not be applied over the carotid sinus nerves, particularly in patients with a known sensitivity to the carotid sinus reflex.
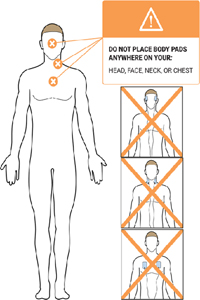
- Stimulation should not be applied over the neck or mouth. Severe spasm of the laryngeal and pharyngeal muscles may occur and the contractions may be strong enough to close the airway or cause difficulty in breathing.
- Stimulation should not be applied transthoracically in that the introduction of electrical current into the heart may cause cardiac arrhythmias.
- Stimulation should not be applied transcerebrally (through the head).
- Stimulation should not be applied over swollen, infected, or inflamed areas or skin eruptions, e.g., phlebitis, thrombophlebitis, varicose veins, etc.
- Stimulation should not be applied over, or in proximity to, cancerous lesions.
- If you have a metallic implant, you may experience pain or discomfort near the implant when applying electrical stimulation, if this should occur discontinue use and seek advice from your doctor
- The electrical stimulation may feel more intense close to a metallic implant. It is safe to continue use provided no pain is experienced. You may need to adjust the intensity to a comfortable level
- During initial treatment phase symptoms may worsen before improving. Your body takes time to adjust to the increased circulation and muscle activity. To reduce this effect, lower the intensity level and the treatment time until your body adjusts. If symptoms continue please consult your doctor
Precautions
- Safety of powered muscle stimulators for use during pregnancy has not been established.
- Caution should be used for patients with suspected or diagnosed heart problems.
- Caution should be used for patients with suspected or diagnosed epilepsy.
- Caution should be used in the presence of the following:
- When there is a tendency to hemorrhage following acute trauma or fracture.
- Following recent surgical procedures when muscle contraction may disrupt the healing process.
- Over the menstruating or pregnant uterus.
- Over areas of the skin which lack normal sensation.
- Some patients may experience skin irritation or hypersensitivity due to the electrical stimulation or electrical conductive medium. The irritation can usually be reduced by using an alternate conductive medium or alternate electrode placement.
- Keep this device out of the reach of children.
- The Advanced Foot Energizer should be used only with the body pads, cords, and accessories recommended for use by the manufacturer.
- Ensure connectors are pressed together firmly with no gap.
![]() DO NOT STAND ON ADVANCED FOOT ENERGIZER – Use only while seated
DO NOT STAND ON ADVANCED FOOT ENERGIZER – Use only while seated